SHANNON, CLARE, IRELAND, March 23, 2026 /EINPresswire.com/ — A newly published study reports that APC-deficient cancer cells may depend on a single metabolic enzyme for survival, revealing a potential strategy for selectively targeting tumours associated with one of the most common mutations in colorectal cancer.
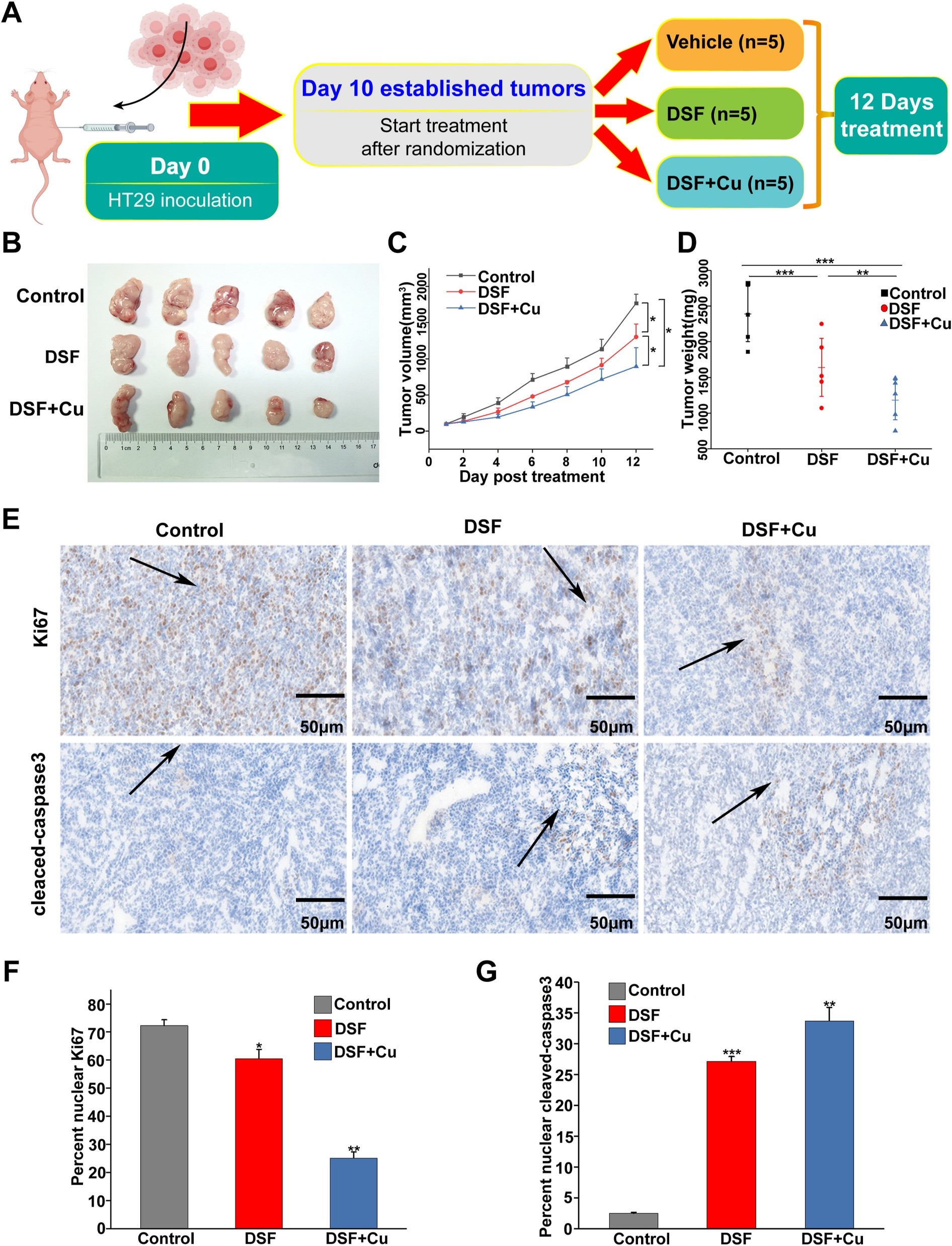
The research identifies ALDH2, an enzyme involved in cellular detoxification, as a critical factor in maintaining viability in cells lacking functional APC. Through a combination of computational screening and experimental validation, the study demonstrates that inhibiting ALDH2 results in a marked reduction in cell proliferation and increased cell death in APC-deficient models.
The underlying mechanism is linked to the accumulation of reactive oxygen species (ROS) following ALDH2 inhibition. This increase in oxidative stress disrupts cellular homeostasis and activates stress-response pathways, including ASK1/JNK signalling, which are known to regulate apoptosis. The resulting shift in apoptotic regulators, including increased BAX and decreased Bcl2, drives programmed cell death in affected cells.
The findings suggest that APC-deficient cells rely on ALDH2 to manage metabolic stress, making them particularly vulnerable when this pathway is disrupted. In contrast, cells with intact APC function show reduced sensitivity to ALDH2 inhibition, highlighting a selective dependency that could be exploited therapeutically.
APC mutations are a defining feature of many colorectal cancers but have remained difficult to target directly. By identifying a downstream metabolic requirement, the study offers an alternative route for intervention that does not rely on directly modifying the genetic mutation itself.
The study also demonstrates that pharmacological inhibition of ALDH2, including with compounds such as disulfiram, can reproduce these effects, supporting the feasibility of targeting this enzyme in a therapeutic context. As an enzyme, ALDH2 represents a more accessible target for drug development compared to many genetic drivers of cancer.
These results contribute to a growing body of work focused on identifying metabolic vulnerabilities in cancer cells. By uncovering a synthetic lethal interaction between APC loss and ALDH2 inhibition, the study provides a framework for developing more targeted treatment strategies.
Further investigation will be required to determine how these findings translate into clinical settings, but the work highlights the potential of exploiting metabolic dependencies to selectively impact cancer cell survival.
# # # # #
Genes & Diseases publishes rigorously peer-reviewed and high quality original articles and authoritative reviews that focus on the molecular bases of human diseases. Emphasis is placed on hypothesis-driven, mechanistic studies relevant to pathogenesis and/or experimental therapeutics of human diseases. The journal has worldwide authorship, and a broad scope in basic and translational biomedical research of molecular biology, molecular genetics, and cell biology, including but not limited to cell proliferation and apoptosis, signal transduction, stem cell biology, developmental biology, gene regulation and epigenetics, cancer biology, immunity and infection, neuroscience, disease-specific animal models, gene and cell-based therapies, and regenerative medicine.
Scopus CiteScore: 7.3
Impact Factor: 6.9
# # # # # #
More information: https://www.keaipublishing.com/en/journals/genes-and-diseases/
Editorial Board: https://www.keaipublishing.com/en/journals/genes-and-diseases/editorial-board/
All issues and articles in press are available online in ScienceDirect (https://www.sciencedirect.com/journal/genes-and-diseases ).
Submissions to Genes & Disease may be made using Editorial Manager (https://www.editorialmanager.com/gendis/default.aspx ).
Print ISSN: 2352-4820
eISSN: 2352-3042
CN: 50-1221/R
Contact Us: editor@genesndiseases.com
X (formerly Twitter): @GenesNDiseases (https://x.com/GenesNDiseases )
# # # # # #
Article Reference
Tingming Liang, Lulu Luo, Yajing Du, Xinbing Yang, Xinru Xu, Haochuan Guo, Zhiheng He, Guang Yang, Li Guo, ALDH2 inhibition induces synthetic lethality in APC-deficient colorectal cancer via ROS/ASK1/JNK pathway, Genes & Diseases, 2026, 102057, ISSN 2352-3042, https://doi.org/10.1016/j.gendis.2026.102057
Genes & Diseases Editorial Office
Genes & Diseases
+86 23 6571 4691
email us here
Legal Disclaimer:
EIN Presswire provides this news content “as is” without warranty of any kind. We do not accept any responsibility or liability
for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this
article. If you have any complaints or copyright issues related to this article, kindly contact the author above.
![]()